Junk DNA: A Journey Through the Dark Matter of the Genome (32 page)
Read Junk DNA: A Journey Through the Dark Matter of the Genome Online
Authors: Nessa Carey

Brittle bone disease isn’t the only human disorder caused by mutations in the untranslated regions at the start of a gene. There is a strong genetic component in about 10 per cent of cases of melanoma, the aggressive skin cancer. A mutation has been identified in some of these genetically driven cases that works in a very similar way to the problem in brittle bone disease. Essentially, a single base change in the untranslated region at the start of a gene creates an abnormal AUG signal in the messenger RNA. This again results in the ribosome starting the amino acid chain too early in the gene sequence. This creates a protein with extra amino acids at the start, which behaves in an abnormal way, increasing the chances of cancer.
6
As always, we need to beware of seeing patterns from too little data. Not all mutations in the untranslated region at the start of a gene create new amino acid sequences. There is another type of skin cancer which is usually much less aggressive than melanoma. This is called basal cell carcinoma, and it too has a strong genetic component. A rare mutation was found in a father and his daughter, both of whom developed this kind of tumour.
The untranslated region at the start of a particular gene usually contains the sequence CGG, repeated seven times, one after the other. The affected father and child had an extra copy of the CGG. Having eight repeats rather than seven predisposed them to basal cell carcinomas. This mutation didn’t change the amino acid sequence of the protein encoded by the gene. Instead, the extra three bases seemed to change the way the messenger RNA was handled by the ribosome, in ways that aren’t very clear. The end result was that the cells of the patients expressed much less of the specific protein than normal.
7
Cancer is a multi-step disease, and although these mutations in the untranslated region at the start of certain genes predisposed the patients to tumours, other events probably also took place in the cells before full-blown cancer developed.
In the beginning was the mutation
But we have already encountered a disorder where an inherited mutation in the untranslated region at the start of a gene leads directly to pathology. This is the Fragile X syndrome of mental retardation (
see page 19
). As a reminder, the mutation is an unusual one. A three-base-pair sequence of CCG is repeated far more times than it should be. Anything up to 50 copies of this repeat is considered to be in the normal range. Fifty to 200 copies is not normally associated with disease, but once the number of repeats gets into this range it becomes very unstable. The machinery that copies DNA for cell division seems to have trouble keeping count of the number of repeats, and even more repeats get added. If this happens in the gametes, the resulting child may have many hundreds or even thousands of the repeats in their gene, and they present with the Fragile X syndrome.
8
The longer the repeat, the lower the expression of the Fragile X gene. As we saw in an earlier chapter, this is because of cross-talk with the epigenetic system (
see page 123
). Where C is followed by G in our genome, the C can have a small modification added to it. This is most likely to happen in regions where this CG motif is present at high concentrations. The large number of CCG repeats in the Fragile X expansion provide exactly this environment. The untranslated region in front of the Fragile X region becomes very highly modified in the patients, and this switches the gene off. Fragile X patients don’t produce any messenger RNA from this gene, and consequently don’t produce any protein from it either.
The effects on the patient of this lack of protein are dramatic. Patients are intellectually disabled but also have symptoms reminiscent of some aspects of autism, including problems with social interactions. Some patients are hyperactive, and some suffer from seizures.
This of course makes us wonder what the protein normally does. The clinical presentation is quite complex, which suggests
that the protein is probably involved in complicated pathways, and this indeed seems to be the case.
As we saw in Chapter 2, the Fragile X protein is usually complexed with RNA molecules in the brain. The protein targets about 4 per cent of the messenger RNA molecules expressed by the neurons.
9
When it binds these messenger RNA molecules, the Fragile X protein acts as a brake on their translation into proteins. It prevents the ribosomes from producing too many protein molecules from the messenger RNA information.
10
This extra level of control on gene expression seems to be particularly important in the brain. The brain is an extraordinarily complex organ, and the cell type that is of most interest to us is the neuron. This is what people usually mean when they talk about brain cells. There are an awful lot of neurons in the human brain, the most recent estimate being just over 85 billion.
11
Each brain contains twelve times as many neurons as there are people on earth. And in the same way that people have complex networks of friends, acquaintances, lovers, families and enemies, neurons are also linked in. What’s startling is the degree of connection between the billions of neurons. Neurons send out projections that connect with other neurons in vast networks, constantly influencing each other’s responses and activities. The precise number of connections is really difficult to estimate, but each cell probably makes at least 1,000 connections with other neurons, meaning our brains contain at least 85 trillion different contact points.
12
It makes Facebook look positively parochial.
Establishing these contacts appropriately is a huge task in the brain. Think of it as arranging to see good friends frequently while trying to avoid the weird guy you met in your first week at college. Contacts are set up and then either strengthened or pruned back, in complex responses to environment and to activities of other neurons in the network. Many of the target messenger RNAs that bind to the Fragile X protein under normal conditions are
involved in maintaining the plasticity of the neurons, allowing them to strengthen and prune connections as appropriate.
13
If the Fragile X protein isn’t expressed, the target messenger RNAs are translated into protein too efficiently. This messes up the normal plasticity of the neurons, leading to the neurological problems seen in the patients.
Researchers have recently shown that they can use this information to treat Fragile X syndrome, at least in genetically engineered animals. Mice which lack the Fragile X protein have problems with their spatial memory, and with their social interactions. A mouse that can’t find its way around and doesn’t know how to react to its fellow mice is a rodent that won’t last long. Researchers used these mice and applied genetic techniques to dial down the expression of one of the key messenger RNAs that would normally be controlled by the Fragile X protein. When they did this, the scientists detected marked improvements in the animals. Spatial memory was better and the mice behaved appropriately around other mice. They were also less susceptible to seizures than the standard Fragile X mouse models.
These symptomatic improvements were consistent with underlying changes that the scientists detected in the brains of the animals.
14
Neurons in normal brains have little mushroom-shaped spines that are characteristic of strong, mature connections. The neurons of humans and mice with Fragile X syndrome have fewer of these, and a larger number of long, spindly, immature connections. After the genetic treatment, there were more mushrooms and fewer noodles.
The most exciting aspect of this was that it suggested it could be possible to improve neuronal function even after symptoms had developed. We can’t use the genetic approach in humans but these data imply that it is worth trying to find drugs that will have a similar effect, as a potential means of treating Fragile X patients. This syndrome is the commonest inherited form of mental retardation
so the benefits of developing a treatment could be dramatic both for individuals and for society.
Now for the other end
As we saw at the start of this book, expansions in a three-base sequence at the other end of a gene can also cause a human genetic disease. The best-known example is myotonic dystrophy, which is caused by expansion of a CTG repeat in the untranslated region at the end of a gene. Repeats of 35 units or above are associated with disease, and the larger the repeat, the more severe the symptoms.
15
Myotonic dystrophy is an example of a gain-of-function mutation. The main effect of the expansion in the Fragile X gene is to stop production of its messenger RNA. But this isn’t the case in myotonic dystrophy. The mutant version of the myotonic dystrophy gene is switched on, resulting in messenger RNA molecules with large expansions at the end of the molecule. It’s these multiple copies of CUG in the messenger RNA (remember that T is replaced by U in RNA) that cause the symptoms. If we turn back to Figure 2.6 (
see page 23
), we can see in outline how this happens. The expanded repeats act like a molecular sponge, soaking up particular proteins that are able to bind to them.
Junk DNA plays a remarkable role in myotonic dystrophy, as shown in Figure 16.4. The CTG expansion in the junk untranslated region binds abnormally large quantities of a key protein.
d
This protein is normally involved in removing the junk DNA that is found between amino acid-coding regions when DNA is first copied into RNA. Because so much of the protein is sequestered onto the expanded myotonic dystrophy untranslated repeat, it can’t carry out its normal function very well. Consequently, lots of RNA molecules from different genes aren’t properly regulated.
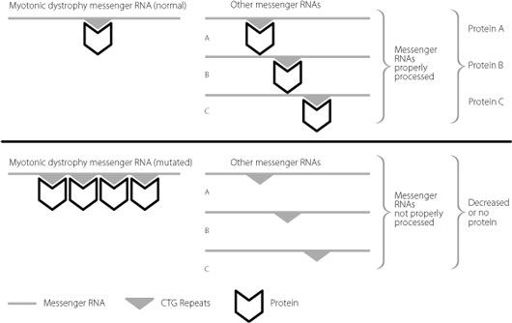
Figure 16.4
The excess binding of proteins to the expanded myotonic dystrophy repeat in the messenger RNA sequesters the proteins away from other RNA molecules that they should also be controlling. The other messenger RNAs are no longer properly processed, and this disrupts production of the proteins that they should be used to produce.
This titration of the binding protein, which occurs in any tissues where both it and the myotonic dystrophy gene are expressed, plays a large role in explaining why the disease can present so differently in different patients. Instead of being all-or-nothing, varying proportions of the binding protein may be ‘left over’ to regulate its target genes. The proportion will depend on the size of the expansion and the relative amounts of myotonic dystrophy messenger RNA and binding protein in a cell.
16
It is worth looking in a bit more detail at the proteins that are ultimately affected by these deficits (proteins A, B and C in Figure 16.4). The best-validated ones are the insulin receptor,
17
a heart protein
18
and a protein in skeletal muscle that transports chloride ions across membranes.
19
Insulin is required to maintain muscle
mass. If the muscle cells don’t express enough of the receptor that binds insulin, they will start to waste away. The heart protein is one that we know is important for the correct electrical properties of the heart.
20
Transport of chloride ions across skeletal muscle membranes is an important stage in the cycles of muscle contraction and relaxation. So, the defects in the processing of the messenger RNAs coding for these proteins are consistent with some of the major symptoms in myotonic dystrophy, i.e. muscle wasting, sudden cardiac death because of fatal abnormalities in heart rhythm, and the difficulty in relaxing a muscle after it has contracted.
Myotonic dystrophy is a great example of the importance of junk DNA in human health and disease. Although the mutation lies in the messenger RNA produced from a protein-coding gene, the mutation has little if any effect on the protein itself. Instead, the mutated RNA region is itself the pathological agent, and it causes disease by altering how the junk regions of other messenger RNAs are processed.
Say ‘AAAAAAAAA’
The untranslated regions at the end of protein-coding messenger RNAs have a number of functions in normal circumstances. One of the most important involves a process that affects all messenger RNA molecules. ‘Naked’ messenger RNA molecules can be broken down in a cell very quickly, via a process that probably evolved to help us get rid of certain types of viruses rapidly. In order to stop this happening, and to make sure the messenger RNA molecules linger long enough to be translated into protein, the messenger molecules are modified very soon after production. Essentially, lots of A bases are added to the end of the messenger RNA, by a process that is outlined in Figure 16.5. There are usually about 250 A bases on the end of a mammalian messenger RNA. They are important for stability and also for making sure that the messenger RNA is exported out of the nucleus where it is made and into the ribosomes where it is translated into protein.
